 On the other hand, when treated as a wave we can predict the position of the electron wave but the momentum cannot be precisely measured. Their speculation about a hard, indivisible fundamental particle of nature was replaced slowly by a scientific theory supported by experiment and. His work evolved the understanding of wave particle duality as when treated as a particle we can easily measure the momentum of a particle but as it is spread out and moving we cannot predict its position. The concept of the atom that Western scientists accepted in broad outline from the 1600s until about 1900 originated with Greek philosophers in the 5th century bce. This relates to the difficult in measuring the quantum values of an electron all at once and was the first time that a scientist had hypothesised that with the precise measurement of one value for example the momentum of the electron, then the position of the electron would be more unknown and less precise. 
His theory went further to also identify that the more precise the position of the electron the less precise the momentum would be. His theory states that there is uncertainty in measuring such features of a particle as the position and momentum of an electron are hard to predict. This helped pave the way for the Manhattan Project, which developed the bombs dropped on Japan. 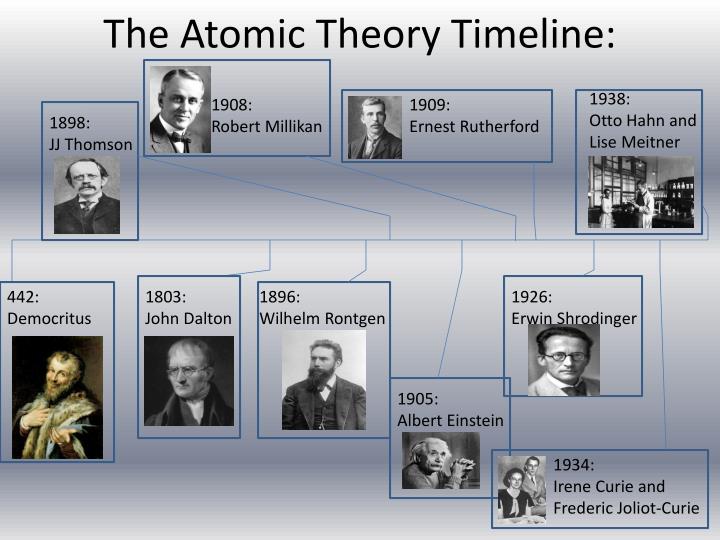 Heisenberg developed a mathematical way of expressing the energy levels of electrons in atoms. Albert Einstein was famously a pacifist, but he urged the US to develop the atomic bomb. Heisenberg’s uncertainty principle followed the work of Albert Einstein and Max Planck in 1927. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
